The Science | THE NUCLEOTIDE EXCISION REPAIR PATHWAY
Targeting the DNA damage response for cancer therapy.
DNA repair and the DNA damage response (DDR) are validated target pathways for therapeutic intervention that have gained increasing interest since the approval of PARP inhibitors in a small subset of human cancers. NERx Biosciences has developed a novel, first in class innovative cancer therapeutic agent that targets the validated DNA damage response pathway. Our targeted therapeutic possesses robust single agent anti-cancer activity and is also designed to work in combination with other clinically approved targeted therapeutics. NERx is positioned to lead first-in-class and first-in-human trials with this novel agent.
The Clinical Reality of Common Cancers
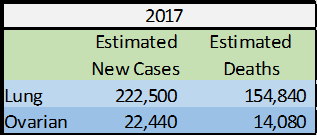
Despite recent advances in immune-oncology, targeted therapeutics and personalized medicine, the clinical reality of many of the most common and most aggressive cancers, including lung and ovarian, remains bleak.
- Lung and ovarian cancer remain difficult to treat cancers, with only a slight decrease in mortality observed over the last 5 years.
- Lung cancer continues to be the number one cancer killer of men and women.
- Greater than 50% of ovarian cancer patients will not be alive 5 years from diagnosis.
- Limited activity and resistance remain a major obstacle with drugs targeting these difficult to treat diseases. Additionally, the low response rate for immune-oncology agents in lung and ovarian cancer limits their widespread use.
There is a high unmet need and significant market opportunity for therapeutics that possess single agent anti-cancer activity AND can be employed in combination with other existing therapies.
Solution: Develop novel biology based targeted therapeutics in a clinically validated pathway.
- NĒRx is targeting DNA repair and the DNA damage response.
- We have developed innovative, biology-based targeted therapies that possess robust single agent anti-cancer activity and are designed to work in specific combination therapeutic regimens.
- These molecules have utility in difficult to treat solid tumors like lung and ovarian.
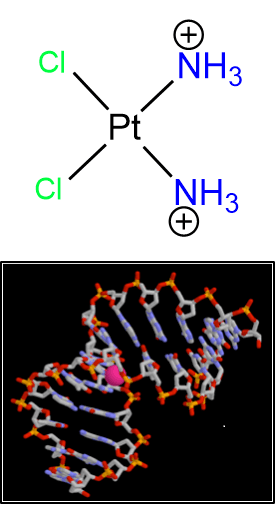
Rationale: biology based targeted therapies designed to work in combination with the most prescribed cancer therapeutic, Cisplatin.
- Cisplatin causes DNA damage
- Cisplatin-DNA damage is repaired by the NER pathway (our namesake).
- Repair of cisplatin-DNA damage is detrimental to efficacy.
- Clinical response to cisplatin is a function of DNA repair.
- Sensitive cancers have reduced repair capacity (testicular).
- Resistant cancers have intrinsically or acquired increased repair capacity (lung and ovarian).
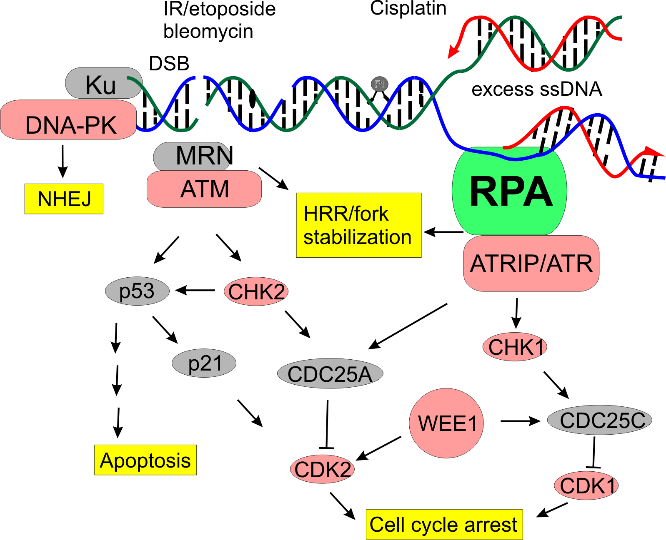
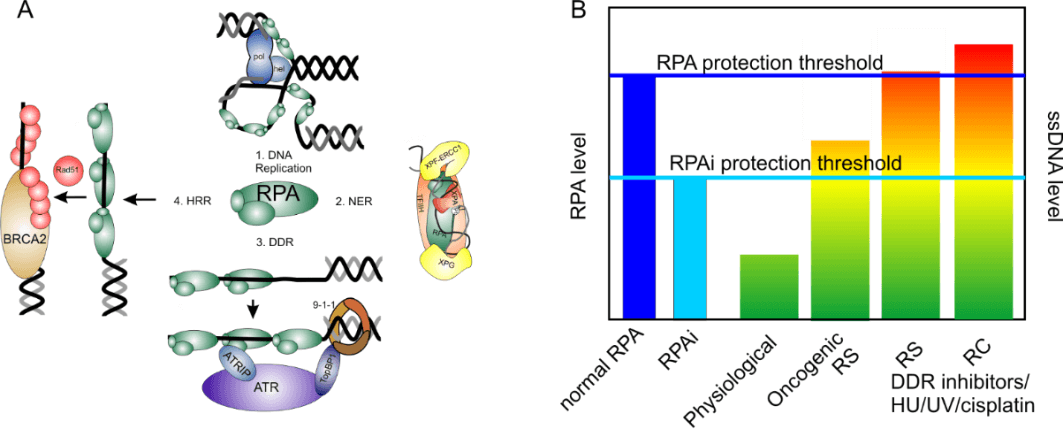
The Target: Develop small molecule inhibitors targeting Replication Protein A (RPA).
- RPA is a novel target within the clinically validated DNA Damage Response pathway.
- Inhibition of RPA role in the Nucleotide Excision Repair (NER) pathway (our namesake) provides enhancement of cisplatin activity.
- Solid tumor reliance on RPA for DNA replication and HRR provides single agent activity.
- Role in DDR presents opportunities for combination with other targeted DDR inhibitors in clinical development.
- NERx molecules target the RPA-DNA interaction; this protein-DNA interaction represents an innovative tactic to treating cancer.
We are specifically targeting difficult to treat cancers, lung and ovarian, that are not effectively treated with existing growth signaling targeted therapeutics and immunotherapy. NERx is positioned to lead the first-in-class and first-in-human trails of novel agents targeting RPA.
Clinical validation
- RPA expression inversely correlates with overall survival in lung and ovarian cancer, providing the clinical rationale for their targeting to treat cancer.